Below are some facts about ethanol production and use and how it relates to your vehicle. Click on the following links to view more topic-specific ethanol facts.
Ethanol and the Economy | Ethanol and the Environment | Ethanol and Energy Security | Food and Fuel Facts
Ethanol Production and Use
The process of ethanol, or ethyl alcohol, production started many decades ago, and the use of ethanol as a transportation fuel has been around since Henry Ford’s Model T. Ethanol is a biodegradeable, high octane fuel that is made from the sugars, starches, and cellulosic matter found in plants. Today, nearly every gallon of gasoline sold in the U.S. contains ethanol, typically a 10% ethanol blend.
Over the past three decades, the ethanol industry in the U.S. has prospered, with more than 200 corn-ethanol biorefineries across the nation with the capacity to produce about 18 billion gallons of clean, renewable fuel. Second generation ethanol plants produce high octane fuel from cellulosic plant matter found in wood chips, corn stalks, and other feedstocks.
There are currently two main processes of traditional corn ethanol production: dry milling and wet milling. Roughly 90% of today’s ethanol industry uses the dry milling process, while the remaining 10% uses the wet milling process.
Dry Milling
In the dry mill process, the whole corn kernel is ground into a flour, or “meal.” The water is then added to the meal to form a “mash,” and enzymes are added to the mash to convert the starch to dextrose, a simple sugar. The mash is then processed in a cooker, cooled, and transferred to fermenters. Yeast is then added and the conversion from sugar to ethanol and carbon dioxide (CO2) begins.
After fermentation, the resulting “beer” is sent to distillation columns where the ethanol is separated from the “stillage.” The ethanol is concentrated through distillation, then dehydrated, and blended with about 2.5% denaturant (such as natural gas) to make it undrinkable and exempt from beverage alcohol tax. The ethanol is then ready for shipment, but the process doesn’t stop there.
The remaining stillage is sent through a centrifuge, separating the grain from the solubles. The solubles are then concentrated through evaporation, resulting in a “syrup.” The coarse grain and syrup are dried together to produce dried distillers grains with solubles (DDGS), a high-protein, high-quality livestock feed. The CO2released during fermentation is captured and sold for use in carbonating soft drinks and to manufacture dry ice.
A Few Dry Milling Facts:
- A dry mill ethanol plant produces 2.8 gallons of ethanol and 17 pounds of valuable feed co-products called distillers grains from just one bushel of corn.
- According to the University of Illinois at Chicago, on a per gallon basis, ethanol plants today require 28% less thermal energy and 32% less electricity than a decade ago.
- Water used to produce a gallon of ethanol has dropped 47% over the last decade.
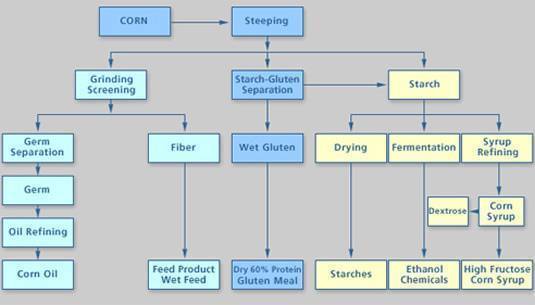
Wet Milling
In the wet milling process, the grain is soaked (or “steeped”) in water and dilluted with sulfurous acid for 24-48 hours to separate the grain into its many component parts. The corn “slurry” is then processed through grinders to separate the corn germ. The corn oil from the germ is then extracted
The remaining fiber, gluten and starch components are further segregated.
The steeping liquor is concentrated in an evaporator, and this “heavy steep water” is co-dried with the fiber component and then sold as corn gluten feed to the livestock industry. The heavy steep water is also sold by itself as a feed ingredient and is used as a component in Ice Ban, an environmentally friendly alternative to salt for removing ice from roads.
The gluten component (protein) is filtered and dried to produce the corn gluten meal co-product. This product is highly sought after as a feed ingredient in poultry broiler operations.
The remaining starch is then fermented into ethanol, using a process very similar to the dry mill process.
Ethanol from Cellulosic Biomass
This diagram shows the basic steps in production of ethanol from cellulosic biomass. Note that there are a variety of options for pretreatment and other steps in the process and that several technologies combine two or all three of the hydrolysis and fermentation steps within the shaded box.
For more information of the ethanol production process, please visit the “How Ethanol is Made” section Renewable Fuels Association (RFA) website by clicking here.
Ethanol and Your Vehicle
- Ethanol is an alcohol primarily produced from corn that contains octane and oxygen, resulting in a fuel that burns more completely and creates less tailpipe emissions.

- Ethanol-blended fuel keeps your fuel system clean for optimal performance because it doesn’t leave gummy deposits. It’s a proven octane enhancer and replacement for lead and other toxic compounds in gasoline. A 10 percent ethanol blend is approved for use by ALL auto manufacturers.
- Ethanol is the highest performance fuel on the market with a 113 octane rating. Several national and international racing teams use ethanol because of its high octane and exceptional performance. NASCAR now runs on E15, and the Indy Racing League has adopted ethanol to replace methanol.
- Ethanol blended fuels, such as the 10 percent blend, are approved for use in small engines, including outboard motors, snowmobiles, lawn mowers, motorcycles and chainsaws. All small engine manufacturers that have tested a 10 percent ethanol blend have approved its use.
- Ethanol guards against gas line freeze by absorbing any moisture that may get in the tank during cold weather, saving consumers money on over-the-counter de-icers.
Unleaded 88/E15: Burns Cleaner. Higher Octane. Better for Your Engine.
- E15 is a blend of 15% ethanol and 85% gasoline. It contains 5% more ethanol than E10, which is the most common fuel used in the U.S. E15 is higher in octane – typically 88 octane while E10 has an octane rating of 87. Retailers are offering E15 as Unleaded 88 to highlight its high octane value.
- Unleaded 88 can be used in all 2001 & newer cars, trucks and SUVs. These model years represent more than 90% of vehicles on the road today. It’s the most tested fuel in history and has no effect on vehicle drivability. To date, more than 15 BILLION miles have been driven using Unleaded 88, and that number continues to grow.
- There is no noticeable difference between the mileage achieved when using Unleaded 88 and the mileage when operating on E10.

- Unleaded 88 is the lowest-cost fuel available for 2001 & newer vehicles, typically selling for 3 – 10 cents a gallon less than E10. The price difference between Unleaded 88 and conventional gasoline without ethanol is around 40 cents.
- Unleaded 88 has not been approved by EPA for use in non-automotive engines like boats, motorcycles, lawn mowers and other small or off-road engines because it wasn’t tested. E10, however, is approved for every on-road and off-road engine.
Ethanol Lowers Gas Prices by $0.50 – $1.50 Per Gallon
According to a study by renowned Energy Economist Philip K. Verleger, consumers are saving $0.50 to $1.50 per gallon on gasoline as a result of ethanol production under the Renewable Fuel Standard (RFS). Verleger served as an advisor on energy issues to both the Ford and Carter Administrations.
“The implication for world consumers is clear… [T]he U.S. renewable fuels program has cut annual consumer expenditures in 2013 between $700 billion and $2.6 trillion,” writes Verleger. “This translates to consumers paying between $0.50 and $1.50 per gallon less for gasoline.”
The study also notes the RFS has added “…the equivalent of Ecuador’s crude oil output to the world market at a time of extreme tightness.” It goes on to note that crude oil prices would be $15 to $40 per barrel higher today without the substantial volumes of ethanol that have been added to petroleum inventories since the enactment of the RFS.